 
Given that:Sublimation energy of K(s) = 89 kJ mol–1Ionization energy of K(g) = 418 kJ mol–1Dissociation energy of Cl2(g) = 244 kJ mol–1Electro attachment energy of Cl(g) = –349 kJ mol–1Formation of KCl(s) = – 437a)-575 kJ mol–1b)-202 kJ mol–1c)-717 kJ mol–1d)-800 kJ mol–1Correct answer is option 'C'. Can you explain this answer?, a detailed solution for Calculate the lattice energy of KCl at 298 K. Besides giving the explanation ofĬalculate the lattice energy of KCl at 298 K. Can you explain this answer? defined & explained in the simplest way possible. 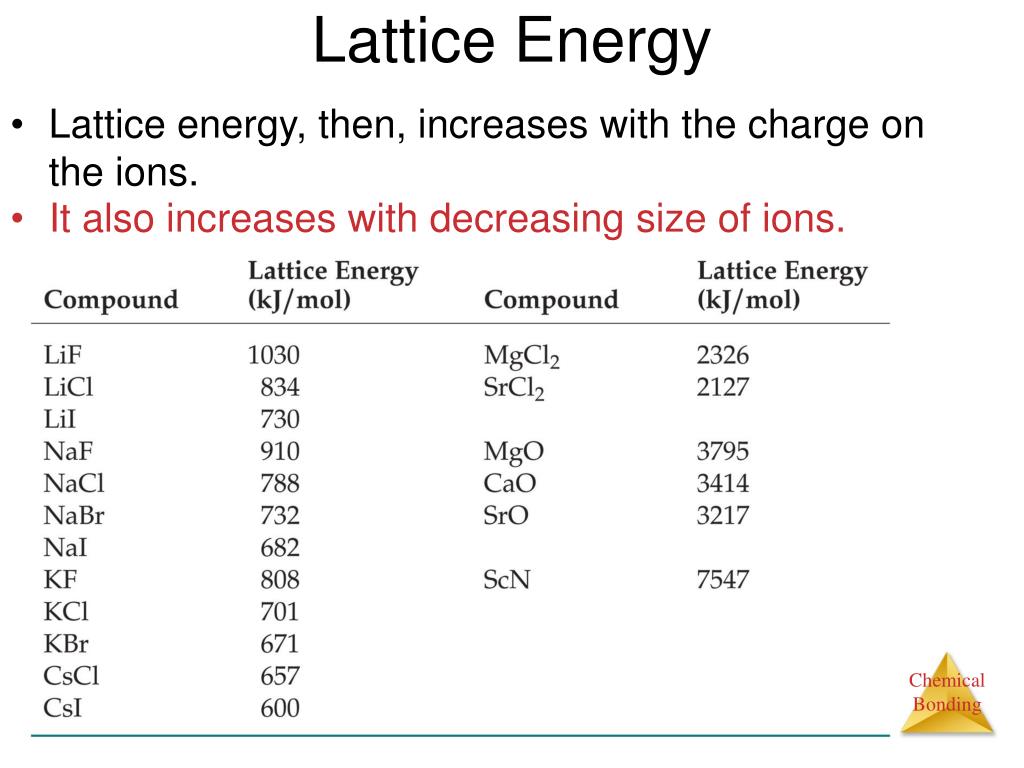
Here you can find the meaning of Calculate the lattice energy of KCl at 298 K. Can you explain this answer? covers all topics & solutions for Chemistry 2023 Exam.įind important definitions, questions, meanings, examples, exercises and tests below for Calculate the lattice energy of KCl at 298 K. Information about Calculate the lattice energy of KCl at 298 K. The Question and answers have been prepared Can you explain this answer? for Chemistry 2023 is part of Chemistry preparation. Therefore, the lattice energy of KCl at 298 K is approximately -7156 kJ mol-1.Ĭalculate the lattice energy of KCl at 298 K. To convert the enthalpy change to the lattice energy per mole of KCl, the Avogadro's number (Na) and the distance between K+ and Cl- ions (r) need to be considered:  
Where ΔHf is the enthalpy of formation of KCl, which can be assumed to be -436.8 kJ mol-1 at 298 K. The lattice energy can be calculated by summing up the enthalpy changes in the cycle: The lattice energy of KCl can be calculated using the Born-Haber cycle: , and the distance between K+ and Cl- ions is 269 pm. , electron affinity of Cl(g) = -349 kJ mol The n values and the electronic configurations (e.c., ionization energy of K(g) = 418.8 kJ mol Where N is the Avogadro's number (6.022x10 -23), and n is a number related to the electronic configurations of the ions involved. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
